The Varicella
Written by: 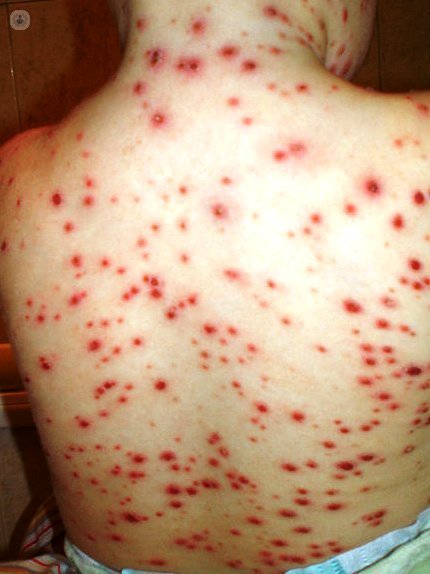 Since February 2016 the varicella vaccine becomes available in Spanish pharmacies after having been withdrawn for nearly three years. In addition, it has introduced in the official vaccination schedule for children 12 to 15 months, risk groups and over 12 years who have not had the disease. Many families lack information or have doubts about vaccination. Other know if your child is properly immunized because they could not complete the second dose by lack of access to the vaccine.
Since February 2016 the varicella vaccine becomes available in Spanish pharmacies after having been withdrawn for nearly three years. In addition, it has introduced in the official vaccination schedule for children 12 to 15 months, risk groups and over 12 years who have not had the disease. Many families lack information or have doubts about vaccination. Other know if your child is properly immunized because they could not complete the second dose by lack of access to the vaccine.
What is Chickenpox?
Chickenpox is a very common infectious disease, especially in childhood, characterized by a very typical vesicular rash that allows clinical diagnosis. It occurs after primary infection with varicella-zoster virus. Humans are the only reservoir. Contagion is fundamentally direct, through droplets emitted by the sick Pflügge or contact with the skin lesions. Infectiousness is very high and the contagious period extends from 1-2 days before the onset of the rash until about 5-7 days after the same. Its clinical course is usually benign, but in a 2 to 6% of cases can cause major complications. If the disease occurs during pregnancy can affect the fetus causing birth defects or chickenpox in the newborn, sometimes even very serious course. , When indicated, etiological treatment is not always effective. Hence the importance of vaccination for primary prevention of disease.
The varicella vaccine
The varicella vaccine virus is attenuated. The usual route of administration is subcutaneous, in the external anterolateral thigh in infants and in the deltoid region in older children, adolescents and adults. In general it is very safe and well tolerated. Adverse reactions are usually mild and occur with a frequency ranging from 5-35% of vaccinees. The most frequent are local reactions such as pain, redness or swelling. Systemic effects are mild fever and rashes that appear between 5 and 30 days after vaccination. Between 3 to 5% of the vaccinated children have localized rash with little more than vesicular maculopapular papulosos elements in the vicinity of the injection site. Another similar percentage may present a generalized rash. In adolescents and adults the frequency of rash reactions may be somewhat higher, especially after the first dose. For practical purposes it is considered to be a prior natural infection with the vaccine when the variceloso rash appears on the first 15 days after vaccination; chickenpox vaccine when it appears between 16 and 42 days; and moderate varicella in vaccinated individuals, if after 42 days appears after vaccination. It is very rare that vaccinated healthy children transmit the virus to susceptible contacts. The transmission has occurred only when the vaccinated person develops exanthema. The incidence of herpes zoster is lower with the vaccinia virus with wild virus. Overall it is estimated that the incidence of herpes zoster in vaccinated children is 4 to 12 times lower than that of unvaccinated children, demonstrating that the vaccine virus is less able to reactivation.
Protective efficacy and number of doses
In Pediatrics administration of a first dose induced seroprotection six weeks in 85% of children between 1 and 12 years amounted to 99.5% after the second dose. In older adults 12 years and seroprotection after the first dose is 80% and 98% after the second dose. It is estimated that the effectiveness of one dose of vaccine in children aged 1 to 12 years is 80-85% for any form of the disease and 95-98% for severe varicella. However, protection against disease with a single dose of vaccine decreases over time and especially after 5 years. It is therefore necessary to administer two doses of vaccine for maximum protective efficacy, the first at 12 and 15 months and the second between 2 and 4 years old.
Indications and contraindications
The varicella vaccine is indicated systematically in susceptible persons aged less than 12 months. Also in susceptible individuals who have been exposed to chickenpox, since vaccination within 3 to 5 days after exposure you can prevent a clinically apparent infection or modify the course of the same. In these cases the recommended regimen is two doses separated by a minimum interval of one month. The vaccine can also be used in certain immunodeficiencies in which, after an individualized assessment, it is assessed that the benefit outweighs the risk.
Regarding contraindications, in addition to general vaccine (anaphylactic reaction to previous dose, severe to any component of the vaccine hypersensitivity, severe acute disease) should be noted: congenital immunodeficiencies (cellular or mixed) and acquired immunodeficiencies (leukemias, lymphomas and malignant) tumors in active phase of the disease; immunosuppressive therapy (up to three months after completion); treatment with high doses of corticosteroids (up to one month after its completion); HIV infection with CD4 + percentage <15% lymphocytes; transplant program that does not meet the conditions to be vaccinated; active tuberculosis.
Vaccination in special situations
- Children who have had chickenpox before the age of 1 year. The vaccination, regardless of history of having had chickenpox in the first year of life, strengthens the protection of those who may have developed an incomplete immunity after natural infection in that period time, interference by maternal antibodies received through the placenta. However, cases can be assessed individually, based primarily on age and safety diagnosis of chickenpox. The infants over 6 months of age suffering from a clinically clear chickenpox, are likely to develop a comprehensive and lasting immunity. They can be considered immune and therefore omit them vaccination. Infants suffering from chickenpox with a lower age 6 months and younger than 1 year in which the disease is mild or very mild, so that his diagnosis of doubt, should be vaccinated at the right age generally , regardless of the aforementioned background.
- Children who report a history of clinically very mild chickenpox and low diagnostic accuracy to standard vaccination age: should be vaccinated according to the normal pattern of two doses.
- Children who have had chickenpox (or shingles) after the administration of the first dose of the vaccine: no need to receive the second dose.
- Children who, on reaching the age of vaccinating susceptible adolescents (12 years) reported having received a single dose before: should receive the second dose.
- Pregnancy. After monitoring of women exposed inadvertently to previously or during the same vaccine it has not been reported no cases of congenital varicella, so it follows that its teratogenicity is very low. However, vaccination is not recommended during pregnancy or the woman becomes pregnant within a month after administration of the vaccine.


